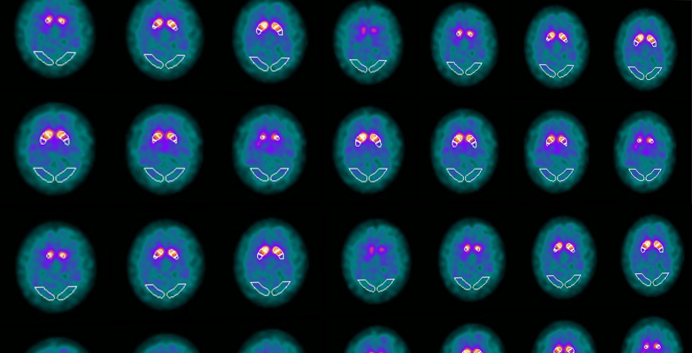
Company will introduce new cDAT™ solution for automated DaTSCAN™ quantification to its platform following CE mark certification to support diagnostics of Parkinson’s disease.
Tampere, Finland, December 22, 2023 — Combinostics, the only company providing a complete imaging and decision support solution spanning the entire patient care pathway for the early detection, diagnosis, and ongoing management of major neurological disorders, today announced that it has obtained CE mark certification under the EU’s Medical Device Regulation (MDR) for its cNEURO cDAT application. The new cDAT application for automated quantification of DAT SPECT images assists in detection of loss of functional dopaminergic neuron terminals in the striatum, which is correlated with diseases associated with dopaminergic deficiencies, such as Parkinson disease or Dementia with Lewy Bodies (DLB).
“cDAT further complements our suite of holistic solutions for neurological disorders” said Lennart Thurfjell, CEO of Combinostics. “With the addition of cDAT we enhance our solution for dementia by providing tools to differentiate between Alzheimer’s disease and Dementia with Lewy Bodies and we extend our offering into the area of movement disorders”.
For more information or to view a demo of Combinostics’ solutions, schedule a demo.
About Combinostics
Combinostics’ AI-powered cNeuro suite of products helps clinicians make a difference in the lives of patients with neurological disorders. By quantifying brain images and integrating patient data from multiple sources with insights from previous patients, the company’s unique software tools provide radiologists and clinicians the support they need for confident, evidence-based diagnostic and management decisions. The company was founded in 2014 and is headquartered in Tampere, Finland. For more information, please visit www.combinostics.com.